|
Measuring Specific Heat
|
|
Objective
|
To enable the student to measure the specific heat of liquids by an electric method |
|
Theory
|
When an electric heater is immersed in a liquid, the electrical energy flowing in heater will be totally converted into heat energy in the liquid. From electricity, the electrical energy is given by E=Pt where "P" is the heater power in watts, "t" time in seconds and "E" electrical energy in joules. From heat, we know
that the heat gained by the liquid is given by H=mc(T2-T1) where "H" is heat energy in joules, "m" liquid mass in "Kg", "c" specific heat in "J/KgC", T1 liquid initial temperature in degrees Celsius before heating, T2 is the liquid temperature after heating in degree Celsius
Assuming there is no heat lost to the surroundings of the liquid, we can say the heat energy gained by the liquid is equal to the electrical energy.
Heat energy gained by liquid=Electrical energy given by heater that is Pt=mc(T2-T1). By solving for "c" we obtain c=pt/(m(T2-T1)). Thus from this equation we need the following data to measure the specific heat of the liquid
- Electric heater power. We can obtain this data from the specification of an electric heater
- Heating time: We need a stop-watch to measure the heating time
- Liquid mass: we need a balance to measure liquid mass
- Initial and final temperature: We need a thermometer to measure temperatures
|
| Tools |
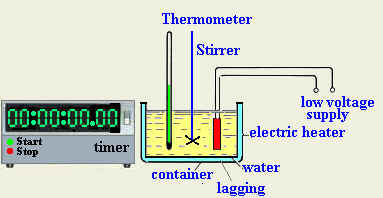
- Container: to hold the liquid
- Liquid: to measure its specific heat
- Lagging: to prevent heat from being lost to the surroundings
- Stirrer: to circulate the liquid and distributed heat evenly so that all the liquid will have the same temperature
|
| Equipment |
- Electric heater: to supply heat
- Thermometer: to measure the rise in the temperature of the liquid
- Stop-watch: to measure time interval during which heat is supplied to liquid
- Balance: measure the mass of the liquid
|
| Precautions |
Heat Precautions
- When heating the liquid, immerse the heater completely in the liquid
- Place the thermometer a way from the electric heater
- Stir the liquid to distribute heat evenly
- Use lagging (heat insulators) to reduce heat loss to surroundings
- The liquid should not reach its boiling point
Thermometer Precautions
- The mercury thread should be kept along the scale to avoid error due to parallax
- line of sight should be perpendicular to scale
- Reading should be at the top of mercury thread
|
|
Procedure and DATA
|
Method-I:
- Prepare all the needed tools and equipment
- Assemble the experiment as shown
- Place a quantity of water in the container
- Place the electric heater in water
- Switch on the heater and the stop-watch
- After several minutes, switch off the heater and the stop watch and record the temperature
- repeat steps 6 and record three readings in the table
- Calculate the average of the three values obtained for the specific heat "c"
|

|
| |
Heater Power "P" |
|
Watts |
|
| |
Mass of water "m" |
|
Kg |
Specific Heat |
| Try |
Stop-Watch time (t) |
Initial-Temperature (T1) |
Final-Temperature (T2) |
c=pt/(m(T2-T1)) |
| 1 |
|
|
|
|
| 2 |
|
|
|
|
| 3 |
|
|
|
|
| |
|
|
Average |
|
|
Method-II:
- Prepare all the needed tools and equipment
- Assemble the experiment as shown
- Place a quantity of water in the container
- Measure the mass of the water
- Place the electric heater in water
- Switch on the heater and the stop-watch
- After each five minutes, record the temperature of water and time
- repeat step "7" for ten times
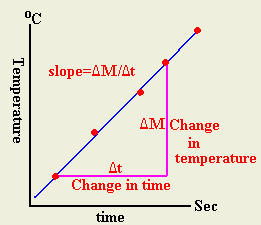
- Plot temperature versus time
- Evaluate the slop from the graph
- Calculate the specific heat of water from slop of the curve
|
|
| |
Heater Power "P" in watts |
|
|
| |
Mass of water "m" in Kg |
|
|
| Try |
Stop-Watch time (t) |
Temperature (T1) |
|
| 1 |
|
|
 |
| 2 |
|
|
| 3 |
|
|
| 4 |
|
|
| 5 |
|
|
| 6 |
|
|
| 7 |
|
|
| 8 |
|
|
| 9 |
|
|
| 10 |
|
|
|
| Analysis |
Questions
- What is the percentage error of your experiment? use %Error=(Actual-Experimental)/Actual
- If your experimental value is less than the actual value, then explain why?
- If your experimental value is greater than the actual value, then explain why?
- Name the objects that will gain heat other than the water?
- Why did we use the electric heater rather than using Bunzen flame?
- Why does the heater should be completely immersed in the water?
- Why should we the use the lagging?
- What is the function of the stirrer?
|
|